9/10/2019 – Co-Founder & Executive Director, David Fajgenbaum, released a memoir about his inspirational battle with Castleman disease and the CDCN’s story—now a national bestseller and available wherever books are sold!







































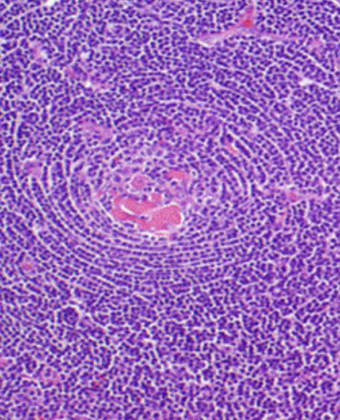
9/10/2019 – Co-Founder & Executive Director, David Fajgenbaum, released a memoir about his inspirational battle with Castleman disease and the CDCN’s story—now a national bestseller and available wherever books are sold!






12/19/2019 – Check out our latest newsletters and CDCN updates here! Sign-up for our newsletter by filling out the form at the bottom of any webpage.

12/21/2019 – Another amazing year for the CDCN has passed. In this news story, we would like to highlight some of our biggest accomplishments of the year. All these accomplishments have only been possible due to the support of hundreds of patients, loved ones, and community members from all around the world.

12/13/2019 – The CDCN presented three Castleman disease-related posters and held a working dinner for hematologists, oncologists, and researchers from around the world at the annual ASH conference in Orlando!

8/26/2019 – Dr. David Fajgenbaum, co-Founder & Executive Director of the Castleman Disease Collaborative Network (CDCN), a physician-scientist at the University of Pennsylvania, and a patient battling the same disease that he studies, were profiled today on Good Morning America as part of a segment about Fajgenbaum’s new book, Chasing My Cure: A Doctor’s Race to Turn Hope Into Action

08/21/2019 – A paper published in the Journal of Clinical Investigation last week describes the treatment of three iMCD patients who previously did not respond to IL-6 blockers like siltuximab, the only FDA-approved drug for iMCD.

7/26/2019 – A clinical trial to evaluate the medication sirolimus for treatment-refractory idiopathic multicentric Castleman disease is launching in Summer 2019! The trial is run by the University of Pennsylvania and the University of Arkansas for Medical Sciences.

7/19/2019 – The Philadelphia City Council unanimously passed a resolution declaring July 23 as World Castleman Disease Day in the City of Philadelphia! This resolution not only recognizes the CDCN’s celebration of the 2nd annual World Castleman Disease Day, but also shows the council’s support in the the continued efforts of the CDCN to accelerate research to find a cure for Castleman Disease and improve the lives of all Castleman patients and their loved ones.

7/17/2019 – Recent research from physicians in the CDCN network has uncovered a gene that is mutated in the lymph node cells of some UCD patients!

6/11/2019 – The Castleman Disease Collaborative Network (CDCN) is excited to announce that we are partnering with the Chan Zuckerberg Initiative (CZI) to accelerate Castleman disease research and further build a prototype that revolutionizes all rare disease research.

6/11/2019 PHILADELPHIA (June 2019) — The Castleman Disease Collaborative Network (CDCN) is proud to announce a new partnership with the Chan Zuckerberg Initiative (CZI) to strengthen and scale the CDCN’s collaborative network approach to advance cures for CDCN and other rare diseases. CDCN’s model inspired CZI’s Rare As One Project, committed to uniting rare disease communities in their quest for cures.

5/3/2019 – Thanks to a study funded by the CDCN’s Castleman Disease Warriors, the Wharton Class of 2015, and the Penn Orphan Disease Center, the first whole genome sequencing of Castleman patients was carried out by Dr. Minji Byun! Dr. Minji Byun, an Assistant Professor of Medicine at the Icahn School of Medicine at Mount Sinai, conducted a research study which had the goal of seeing if a possible cause of CD could be a genetic defect.

4/18/2019 – CDCN leaders David Fajgenbaum (Co-Founder), Mary Zuccato (COO), and Dustin Shilling (Senior Scientific Advisor) published “The Collaborative Network Approach: A model for advancing patient-centric research for Castleman disease and other rare diseases” in a 2019 review article in the journal Emerging Topics in Life Sciences. This paper outlines the CDCN’s novel strategy to accelerate research and reach the goal of finding an effective treatment for all Castleman disease patients.

12/21/2018 – This has been an amazing year for the CDCN! In honor of all the progress that we have made thanks to support of hundreds of patients, loved ones, and community members from around the world, we wanted to highlight some of our biggest accomplishments of 2018. Check out our top ten highlights below!

12/18/2018 – The Castleman Disease Collaborative Network (CDCN) is excited to announce the recent publication of a study that identified key laboratory markers that help to predict whether patients with idiopathic multicentric Castleman disease (iMCD) will respond to siltuximab.

12/18/2018 – The Fourth Annual Quest for a Cure Gala was an incredible success! Quest for a Cure was held on November 16, 2018 at the beautiful Arts Ballroom in Philadelphia, PA. Our CDCN community of patients, loved ones, and supporters came together for a beautiful evening filled with cocktails, hoeur d’oeuvres, a seated dinner, and a silent auction.

12/7/2018 – The Castleman Disease Collaborative Network had its best showing yet at the American Society of Hematology’s Annual Meeting and Exposition, the premier event of the global hematology community! This year, ASH’s annual meeting brought together over 27,000 researchers, physicians, and educators from all over the world, including many of the world’s foremost experts in Castleman disease.

10/8/2018 – The 2018 Patient and Loved One Summit was a hit! The fifth annual summit took place on September 21-22, 2018 at the University of Pennsylvania and brought together over 90 patients, loved ones, and CDCN volunteers and staff from around the world.

9/28/2018 – The CDCN convened 42 experts from 10 different countries to establish the first-ever treatment guidelines for idiopathic (HHV-8-negative) multicentric Castleman Disease (iMCD) based on data from over 300 iMCD patients.

8/13/2018 – A new French reference center for Castleman disease held a launch meeting in Paris, “Premiere Journee du Centre de Référence ‘Maladie de Castleman!” The meeting was led by the Director of the new Center, Dr. Eric Oksenhendler. Dr. Oksenhendler has spent over 20 years pushing the Castleman disease field forward, including a large case series from as far back as 1996 that highlighted the significant morbidity and mortality associated with multicentric Castleman disease at the time.

6/20/2018 – We are proud to announce the publication of our SPEED I (Serum Proteomics Evaluation for Etiology and Pathogenesis Data I) research study in the American Journal of Hematology in April 2018 titled Plasma proteomics identifies a ‘‘chemokine storm’’ in idiopathic multicentric Castleman disease. The study found that iMCD is not simply a disorder of IL-6; it’s a complex inflammatory disorder that the global medical community is just beginning to unravel.

3/28/2018 – The CDCN is excited to announce funding for four high-impact research proposals! Each research project was awarded an individual grant of $42,000. The grants were awarded to Dr. Minji Byun at the Icahn School of Medicine, Dr. Robert Ohgami at Stanford University, Dr. Pillai at the Children’s Hospital of Philadelphia, and Dr. Wenbin Xiao at Memorial Sloan Kettering Cancer Center.

1/30/2018 – The TODAY aired a special on Dr. Fajgenbaum and his remarkable story called “Doctor with rare disease racing to save his own life.” The segment highlighted the challenges faced by rare disease and Castleman disease patients, as the medical community often has poorer understandings of how rare diseases work. Watch the story here!

12/1/2017 – Co-Founder and Executive Director of the CDCN, Dr. Fajgenbaum, traveled to New York City to share his story at the Forbes Healthcare Summit. Watch his spotlight entitled “Treating My Own Cancer” here!

4/11/2017 – Dr. David Fajgenbaum, Co-Founder of the CDCN and a Castleman disease patient, was featured by the NYTimes and Huffington Post. Check out the stories here!

4/11/2017 – The CDCN announced a collaboration with Janssen Research & Development, LLC, to conduct a proteomics study of idiopathic multicentric Castleman disease (iMCD).

2/28/2017 – Big strides were made recently for the diagnosis of Castleman disease patients! The first-ever diagnostic criteria for HHV-8-negative “idiopathic” Multicentric Castleman disease has been published in the top hematology journal, Blood. Though Castleman disease was first described in 1954, this is the first time there has ever been a diagnostic criteria for it.

11/28/2016 – Castleman Warriors Mileva Repasky, Shannon Baker and Dr. David Fajgenbaum were featured in the November 28, 2016 edition of CNS News!

6/30/2016 – Dr. David Fajgenbaum answered the top questions asked by patients and provided an update on how our research is answering your questions in the CDCN’s first webinar on August 29th, 2016. Watch the webinar here!

6/26/2016 – The CDCN has made a lot of progress on its mission! Multiple promising studies into Castleman disease are under way, fundraising and patient outreach events have been a huge success with more on the way, and numerous papers have been published in high-impact journals.

6/6/2016 – Dr. Alexander Fossa is providing essential samples for several important studies which hope to identify the cause, key cell types, and a diagnostic test for Castleman disease. Dr. Fossa provided the most samples for the HUNT study, control samples for HUNT II, and serum samples from for SPEED II.

3/20/2016 – CDCN team members recently co-authored four papers in highly-regarded international journals, including two this week in the Lancet Hematology. One Lancet Hematology paper is the largest and most in-depth study of HHV-8-negative (idiopathic) multicentric Castleman disease.

12/1/2015 – The CDCN and its high-impact research programs have been profiled in several news media outlets recently including ABC-11, Fox 29, and the Philadelphia Inquirer, and was featured at the Medidata Congress!

8/25/2015 – Led by student leaders in the Class of 2015, the Wharton Business School community rallied together to do their part to take down Castleman disease (“Wharton Knock Out Castleman Disease Campaign” led by Andrew Towne) by raising enough funds to enable a top-priority research study!

6/12/2015 – Raj, a Castleman disease patient, has fought back against Castleman disease by providing tissue samples for research and raising enough funds ($24,000) to pay for an entire study! Six other patients also contributed blood samples for the new serum proteomics study.

3/6/2015 – On February 27-28, 2015, the CDCN co-hosted the first-ever International Castleman Disease Pathology Preceptorship at the University of Pennsylvania. The Preceptorship included the eight top hematopathologists from across Latin America and the faculty included Drs. David Fajgenbaum, Dale Frank, and Adam Bagg from from the University of Pennsylvania as well as Dr. Ahmet Dogan from Memorial Sloan Kettering Cancer Center.
1/7/2015 – Dr. David Fajgenbaum, co-Founder & Executive Director of the CDCN and Adjunct Assistant Professor of Medicine in the division of Hematology/Oncology at the University of Pennsylvania, was named to Forbes Magazine’s 30 Under 30 list in healthcare! Dr. Fajgenbaum is a physician-researcher-entrepreneur that has repeatedly used personal adversity as motivation to create groundbreaking initiatives that overcome serious problems.

11/21/2014 – CDCN co-founders, Drs. David Fajgenbaum and Frits van Rhee, presented their new model of pathogenesis and classification system at HEMO, the Brazilian Congress on Hematology and Hematotherapy, in Florianopolis, Brazil. HEMO is the fourth largest hematology conference in the world.

5/8/2014 – Researchers with the CDCN develop a new disease classification system and propose a model of pathogenesis for the potential causes of idiopathic MCD, and provide a new clarity of insight that will expedite further research into the diagnosis and treatment of Castleman disease.

7/14/2014 – CDCN Board president, Greg Pacheco, shares his story about his battle with Castleman disease. He and his wife, Charlyn Pacheco, established the nonprofit organization called CARE (Castleman’s Awareness and Research Effort) in 2007 and raised enough money to fund the Greg Pacheco Castleman Research Laboratory at UAMS. In 2012, Greg partnered with Dr. David Fajgenbaum to establish the Castleman Disease Collaborative Network.

7/10/2014 – The Castleman Disease Collaborative Network (CDCN) is fighting to cure Castleman disease through a first-of-its-kind model. Rather than simply contributing funds into the existing biomedical research system, the CDCN has decided to take a more systematic, coordinated approach.

6/10/2014 – The U.S. Food and Drug Administration today approved Sylvant (siltuximab) to treat patients with multicentric Castleman disease (MCD), a rare disorder similar to lymphoma (cancer of the lymph nodes).